1
"Substrate elasticity controls fibroblast motility on non-oxidized PDMS under weak adhesion" by Arata Nagai, Kyotaro Kanazashi, Hiromu Kuwabara, Yuuta Moriyama, Toshiyuki Mitsui is published in BPPB as the J-STAGE Advance Publication.
2026 May 09 BPPB
A following article is published as the J-STAGE Advance Publication in "Biophysics and Physicobiology".
Arata Nagai, Kyotaro Kanazashi, Hiromu Kuwabara, Yuuta Moriyama, Toshiyuki Mitsui
"Substrate elasticity controls fibroblast motility on non-oxidized PDMS under weak adhesion"
URL:https://doi.org/10.2142/biophysico.bppb-v23.0016
- Abstract
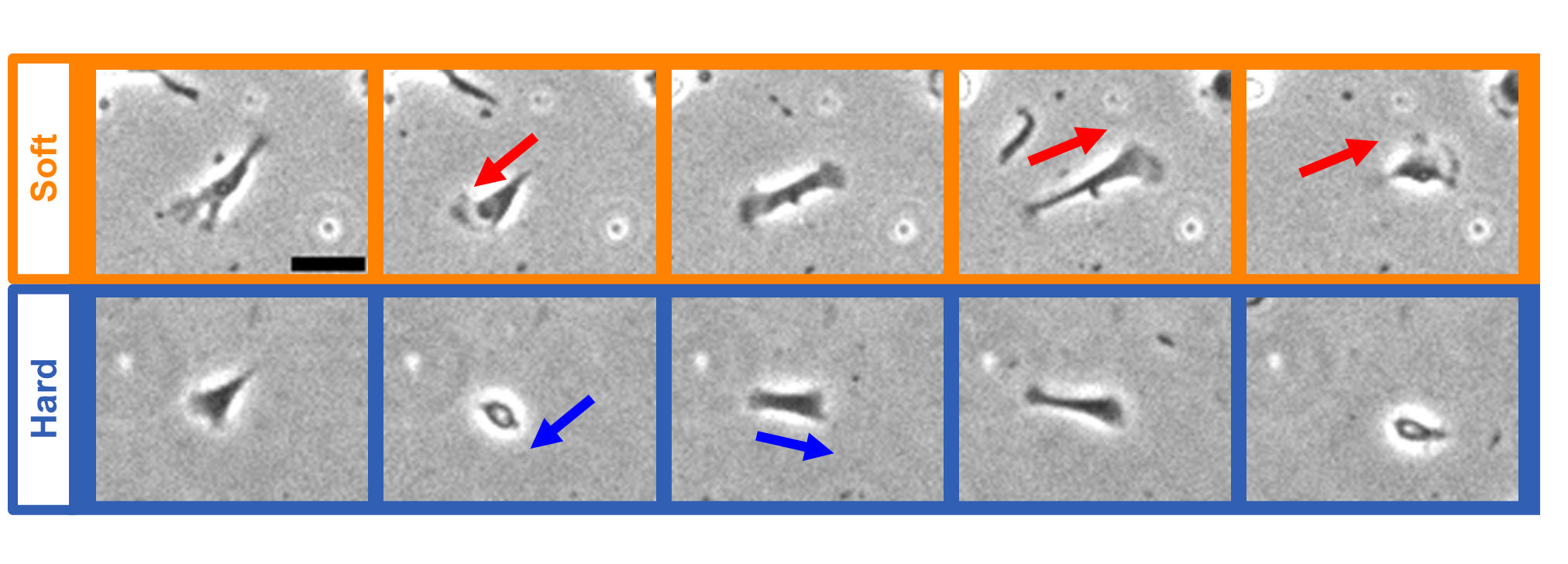
- Cardiac fibroblasts play a pivotal role in heart tissue dynamics, responding to mechanical and interfacial cues in their microenvironment. This study examines how substrate stiffness and surface condition influence the motility and morphology of chicken embryonic cardiac fibroblasts cultured on collagen-coated polydimethylsiloxane (PDMS) substrates with approximate elastic moduli of ~10 kPa and ~400 kPa, representing mechanically soft and stiff conditions, respectively. The PDMS surfaces were used without plasma or UV/ozone treatment, retaining their native hydrophobic character without surface oxidation. This absence of surface activation resulted in weak cell-substrate adhesion, leading to delayed spreading and frequent partial detachment during migration. Using time-lapse phase-contrast microscopy, we observed irregular motility cycles characterized by contraction-driven retraction, followed by partial detachment and re-adhesion. On soft substrates, fibroblasts exhibited longer motility cycles and left trailing edges upon retraction, while on stiff substrates, cells remained rounded and displayed less polarized motion. Quantitative analysis revealed that the duration of static phases and the degree of cell elongation varied with substrate stiffness but were also strongly influenced by adhesion instability due to unoxidized PDMS surfaces. These findings clarify the roles of substrate stiffness and surface adhesion in regulating fibroblast motility, and demonstrate that non-oxidized PDMS surfaces—though less adhesive—can preserve a rounded, less-spread cell morphology. Rather than promoting full spreading, such conditions may maintain fibroblasts in a mechanically less engaged or undifferentiated state. This work provides insight into how surface condition and mechanics together shape fibroblast behavior, with implications for mechanobiology and in vitro model design using non-activated biomaterials.