男女共同参画学協会連絡会
支援企業による広告記事
- ソーラボジャパン株式会社
- CAD を使って光学装置を設計してみよう
- 「生物物理」2025年10月号
- ソーラボジャパン株式会社
- 次世代2光子顕微鏡―小型化がもたらす新たな可能性
- 「生物物理」2024年10月号
- ソーラボジャパン株式会社
- サイエンティフィックカメラと周辺機器の同期
- 「生物物理」2023年10月号
- ソーラボジャパン株式会社
- 顕微鏡のリノベーション ~ 顕微鏡ポートを活用した光学系の導入
- 「生物物理」2022年12月号
「Biophysics and Physicobiology」に Kosuke Morikawa, Takuji Oyama, Masayuki Oda による "Revisiting hydrolytic mechanisms of bacterial RNase HI: Towards clarification of single mobile Mg2+ action in nuclease reactions" をJ-STAGEの早期公開版として掲載
2026年04月18日 学会誌
日本生物物理学会欧文誌[Biophysics and Physicobiology]に以下の論文が早期公開されました。
Kosuke Morikawa, Takuji Oyama, Masayuki Oda
"Revisiting hydrolytic mechanisms of bacterial RNase HI: Towards clarification of single mobile Mg2+ action in nuclease reactions"
URL:https://doi.org/10.2142/biophysico.bppb-v23.0014
- Abstract
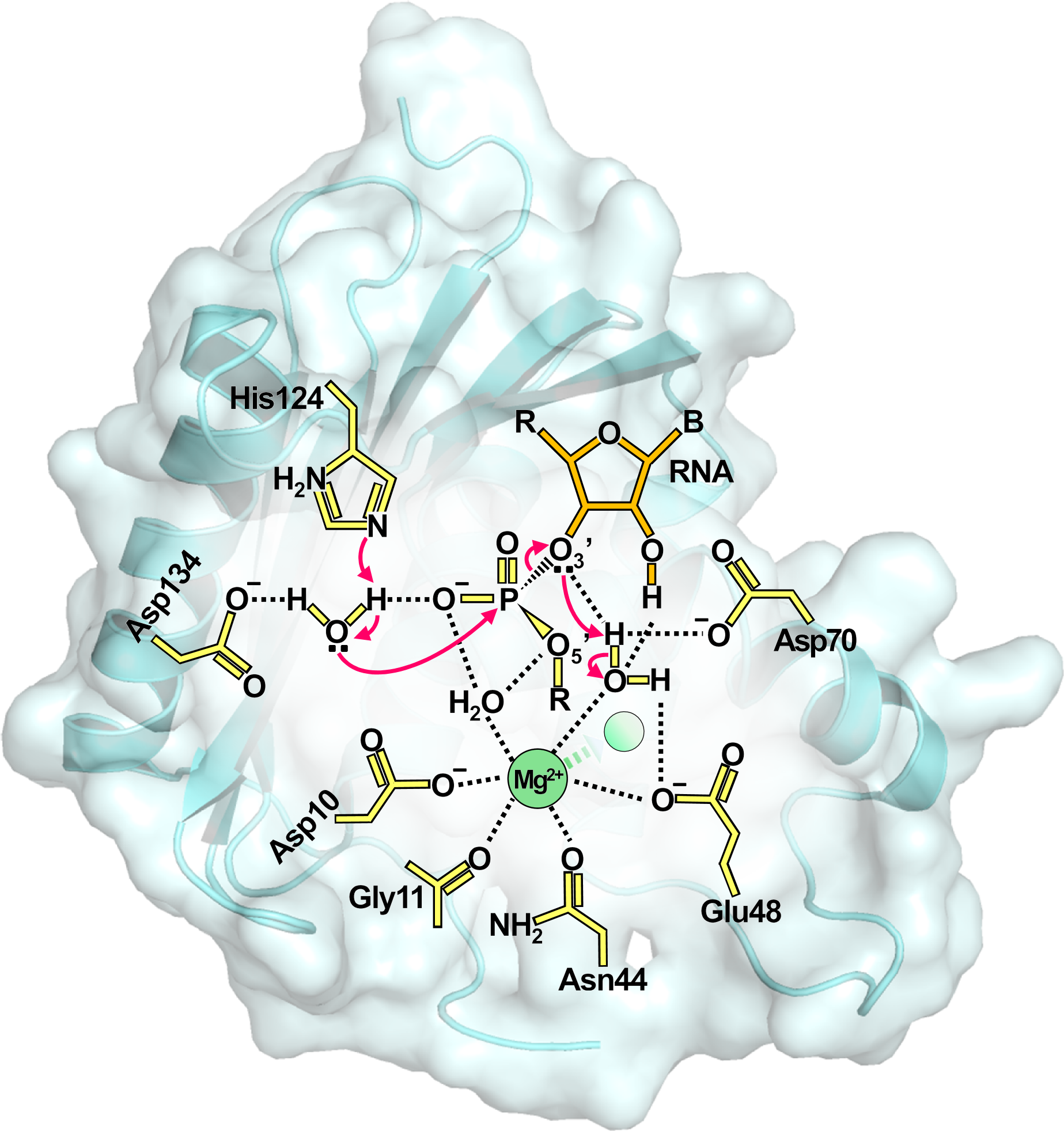
- Cellular nuclei contain various nucleases that play major roles in gene expression and regulation. They are classified into various categories based on their three-dimensional structures and specificities. Since the discovery of ribozyme in the early 1980, one nuclease class has attracted significant attention. The cleavage reactions of these nucleases essentially require Mg2+ cations and produce a 5'-phosphate nucleotide and a 3'-nucleoside at the cleavage site of polynucleotides, similarly to the reaction of ribozyme. In particular, Drs. Thomas A. Steitz and Joan A. Steitz proposed an attractive and general model for the phosphoryl transfer reaction. This was later named the “two-metal-binding mechanism”. In accordance with recent developments in cryo-electron microscopy (cryo-EM), many structural reports published about Mg2+-dependent nucleases or spliceosomes support the “two-metal-binding mechanism” for hydrolytic schemes. However, the direct visualization of Mg2+ on electron-density maps requires high resolution analyses, regardless of X-ray crystallography or cryo-EM. Furthermore, recent biochemical studies combined with structural data support the similarity in hydrolytic mechanisms between ribonuclease HI (RNase HI) and restriction endonucleases. Since the late 2000s, molecular dynamics approaches have also appeared to support the hydrolytic mechanism of single mobile metals. This review aims to critically reconsider Mg2+-dependent hydrolytic mechanisms, with a particular focus on bacterial RNase HI.
URL: https://doi.org/10.2142/biophysico.bppb-v23.0014